
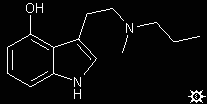
#23. 4-HO-MPT
TRYPTAMINE, 4-HYDROXY-N-METHYL-N-PROPYL; 4-INDOLOL, 3-[2-(METHYLPROPYLAMINO)ETHYL]; 4-HYDROXY-N-METHYL-N-PROPYLTRYPTAMINE; 3-[2-(METHYLPROPYLAMINO)ETHYL]-4-INDOLOL
|
| [3D .mol Image] |
To 8 mL of a stirred solution of LAH (1 M in THF under N2), there was added dropwise a solution of 0.48 4-acetoxyindol-3-yl-N-methyl-N-propylglyoxylamide in 8 mL anhydrous THF. When the addition was complete, the reaction mixture was brought to a reflux for 15 min. After cooling to 40 °C, sufficient water was added to decompose both the reaction complex and the excess hydride. After filtration through Celite (under an N2 atmosphere), the solvent was removed under vacuum, and the oily residue dissolved in MeOH, neutralized with HCl, and Et2O added until crystallization started. Thus there was obtained 0.23 g (54% of theory) 4-hydroxy-N-methyl-N-propylindole hydrochloride (4-HO-MPT) with a mp 162-163 °C. Anal: C,H,N.
DOSAGE : unkown
DURATION : unknown
QUALITATIVE COMMENTS : (with 8 mg, orally) "There was a very mild visual distortion, and a prominent vertigo without nausea. In the second hour there is still some enhancement of visual detail, but I am not endowed with the flight of ideas of philosophical concepts as with psilocin. I am rapidly subsiding and I am able to eat normally. Residual insomnia lasted eight hours."
EXTENSIONS AND COMMENTARY : As with the discussion presented with the lower homologue, 4-HO-DET, there is not enough here to give a fair estimate of either dose or duration. In my file under this compound I can fine only this one report. Extrapolation suggests that this might be yet another material active in the psilocin range of somewhere up to 20 milligrams, all orally. Maybe it is a generality that anything with up to six carbons attached one way or another to the tryptamine nitrogen atom (and all sporting a 4-hydroxy group, of course) will be active in the 10 to 20 milligram range. This certainly holds for the methyl-methyl, methyl-ethyl, methyl-propyl, methyl-isopropyl, diethyl, dipropyl and diisopropyl. That makes it a pretty good generalization.
How far can this argument be pushed? What about one of the N-alkyl groups having four carbons? Keeping the other N-alkyl group as the smallest and most simple methyl group, all four isomeric compounds are known. There is the n-butyl isomer (4-HO-MBT, an oil), the isobutyl isomer (4-HO-MIBT, mp 142-145 °C), the secondary butyl isomer (4-HO-MSBT, mp 138-140 °C) and the tertiary butyl isomer (4-HO-MTBT, mp 225-226 °C). Of these four materials only 4-HO-MTBT has been looked at as a possible psychedelic. Some 15 milligrams produced virtually no effects, maybe a hint of something in a few minutes and then nothing. Probably pure placebo.
Many yet heavier substitution patterns are in the literature but they, too, are unexplored. The symmetrical disubstituted isomers are listed separately in these recipes.